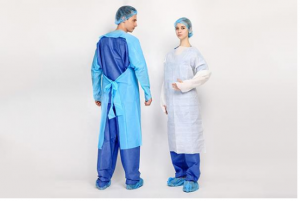
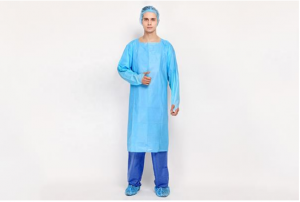
Songjiang, Shanghai Apr 15, 2026 (Issuewire.com) - Following a successful showcase of professional expertise in protective apparel and protective innovation, CHONGJEN, a leading Custom Durable CPE Gowns Manufacturer, has concluded its successful participation at Hospitalar 2025. The event served as a critical platform for the company to demonstrate its advanced Chlorinated Polyethylene (CPE) protective solutions to the Latin American medical community. These specialized gowns, which are a cornerstone of the company’s manufacturing portfolio, feature a thumb-loop design to ensure sleeve stability and are engineered from high-density materials to provide a superior fluid barrier. As a manufacturer capable of customizing thickness, sizing, and tensile strength, the organization highlighted how these durable gowns meet the specific ergonomic and safety demands of frontline healthcare workers in high-risk environments.
Global Healthcare Industry Outlook and PPE Market Trends
The global landscape for medical disposables and personal protective equipment (PPE) is undergoing a significant transition toward higher quality standards and supply chain transparency. In the years following global health crises, the market has matured beyond emergency procurement, with a renewed focus on long-term safety infrastructure and material science innovation. According to industry analysis, the demand for specialized isolation apparel is projected to grow steadily as healthcare systems in emerging markets, particularly in Latin America and Southeast Asia, expand their surgical and intensive care capacities.
One of the defining trends in the industry is the shift toward Chlorinated Polyethylene (CPE) over traditional low-density polyethylene (LDPE). CPE offers superior flexibility and puncture resistance, making it the preferred choice for environments where healthcare workers are at risk of fluid exposure. Furthermore, there is an increasing demand for "tailored" protection. Medical institutions are no longer satisfied with one-size-fits-all solutions; they require manufacturers who can adapt product specifications to the physical requirements of their workforce and the specific risks of their clinical environments.
Regulatory compliance has also become a central pillar of the industry. With the implementation of more rigorous standards for medical devices globally, manufacturers must demonstrate total control over their production processes. This has favored vertically integrated companies that manage everything from film blowing to final assembly. As hospitals prioritize environmental and fiscal sustainability, the durability of disposable products has become a key metric, as higher-quality gowns reduce the likelihood of barrier failure and the subsequent costs associated with healthcare-acquired infections.
Hospitalar 2025: A Strategic Gateway to the Latin American Market
Hospitalar 2025, held at the São Paulo Expo in Brazil, served as the most significant gathering of medical professionals and healthcare suppliers in the Americas. As a premier trade fair, it attracted thousands of exhibitors and tens of thousands of visitors, including hospital administrators, distributors, and government health officials. The event is recognized as the primary platform for introducing new medical technologies and consumable products to the South American market, a region currently witnessing rapid modernization of its healthcare infrastructure.
The 2025 edition of the fair emphasized the themes of efficiency and professional safety. For international manufacturers, the event provided a critical opportunity to engage with regional distributors and understand the nuances of the Brazilian medical market, including the specific requirements of the National Health Surveillance Agency (ANVISA). The halls of the São Paulo Expo facilitated a high level of technical exchange, allowing participants to evaluate the latest advancements in protective materials against the practical needs of regional hospitals.
During the exhibition, the focus remained on the reliability of the medical supply chain. Discussions frequently centered on the need for consistent product quality in high-volume consumables. For an organization specializing in durable protective apparel, Hospitalar offered a unique environment to demonstrate product integrity through physical samples and technical documentation. The event allowed for direct feedback from end-users, which is essential for refining product designs to better suit the intense operational demands of Latin American clinical settings. The networking sessions conducted during the fair underscored a robust demand for direct-to-factory partnerships to ensure stable pricing and quality in the region.
CHONGJEN: Core Strengths, Product Applications, and Market Presence
Established as a manufacturing and trading entity headquartered in Shanghai, CHONGJEN has spent over a decade developing a comprehensive portfolio of healthcare and personal protection solutions. The organization operates on a foundation of technical expertise and a commitment to building long-term partnerships through consistent product delivery.
Manufacturing Advantages and Technical Capabilities
The organization’s core strength lies in its integrated production model. By maintaining advanced manufacturing facilities, the company ensures that every product, from disposable gloves to isolation gowns, adheres to stringent quality control protocols.
Specialized Material Production: The facility utilizes professional film blowing and rolling machines to produce high-quality CPE and TPE films, ensuring the raw materials meet precise density and strength specifications.
Customization and OEM Services: Recognizing the diverse needs of global clients, the company provides extensive OEM services. This includes custom sizing, specific gram weights (GSM) for different levels of protection, and customized branding for international distributors.
Comprehensive Quality Systems: All manufacturing processes are monitored to ensure compliance with international safety standards, providing the necessary documentation for export to highly regulated markets in Europe, North America, and Australia.
Primary Product Application Scenarios
The product range is designed for versatility across multiple high-stakes industries. The durable CPE gowns and related disposables are utilized in:
Medical and Healthcare: Serving as primary isolation barriers in emergency rooms, dental clinics, and surgical wards to prevent cross-contamination.
Food Processing and Service: Ensuring hygiene standards in meat, dairy, and beverage production facilities where fluid resistance is essential for worker safety and product purity.
Industrial Manufacturing: Protecting personnel in cleanrooms and electronic manufacturing environments from dust, splashes, and particulate matter.
Laboratory Research: Providing chemical-resistant barriers for technicians handling biological samples or sensitive reagents.
Major Client Portfolios and Case Success
The company’s market reach extends to over 20 countries and regions, including significant footprints in the United States, the European Union, and Southeast Asia. The client base primarily consists of large-scale medical supply distributors and industrial wholesalers who require high-volume, consistent production. By acting as a reliable supply chain partner, the manufacturer has supported numerous hospital networks in optimizing their PPE procurement. Case studies from long-term partners highlight the company's ability to maintain stable supply even during periods of market volatility, as well as its responsiveness in adjusting product designs based on field feedback. This collaborative approach has resulted in products that are not only protective but also ergonomically suited for extended use in professional environments.
Conclusion
The successful engagement at Hospitalar 2025 marks another milestone in the company’s mission to advance global safety standards. By combining advanced material science with a deep understanding of the practical needs of healthcare providers, the organization continues to reinforce its reputation as a Leading Manufacturer of Custom & Durable CPE Gowns. The future of the protective products industry lies in the ability to provide reliable, high-performance solutions that are tailored to the evolving challenges of modern medicine and industry. Through continuous investment in manufacturing technology and a steadfast commitment to quality, the company remains dedicated to protecting those on the front lines of global health.
For further information regarding the product portfolio and manufacturing capabilities, please visit the official company website: https://www.chongjen.com/
Media Contact
Shanghai CHONGJEN Industry Co., Ltd. info@chongjen.com https://www.chongjen.com/