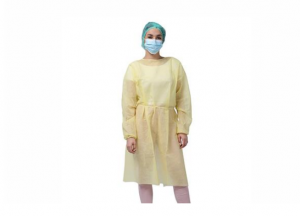
Songjiang, Shanghai Apr 15, 2026 (Issuewire.com) - The global demand for high-integrity barrier protection has reached a pivotal juncture, where clinical efficacy must be met with supply chain reliability. As a Professional Chinese Economical Isolation Gown Supplier, CHONGJEN has established a benchmark in the manufacturing of disposable protective apparel, offering an essential line of defense for medical and industrial personnel. These isolation gowns are meticulously crafted from specialized non-woven materials, including Spunbond Polypropylene (PP) and PE-coated fabrics, designed to offer superior resistance against fluid penetration, particulate matter, and cross-contamination. By integrating high-performance protection with cost-effective manufacturing processes, the organization ensures that healthcare facilities can maintain rigorous safety protocols without exceeding budgetary constraints. The gowns feature ergonomic designs, such as knit or elastic cuffs and secure neck closures, providing both comfort and comprehensive coverage in high-risk environments.
Global Industry Trends: The Strategic Shift in Medical Protection
The landscape of the medical protective equipment industry has undergone a radical transformation over the last decade. Historically, the procurement of personal protective equipment (PPE) was viewed through a purely transactional lens. However, the current global trajectory shows a definitive shift toward strategic, long-term partnerships with manufacturers who can guarantee transparency and compliance. Market analysts project a sustained growth in the disposable medical supplies sector, driven by heightened awareness of hygiene standards, an aging global population, and more stringent occupational safety regulations in regions ranging from North America to Southeast Asia.
One of the most significant trends in the industry is the professionalization of the supply chain in China. Manufacturers are no longer merely assembly hubs; they have evolved into research-driven centers of excellence. There is an increasing focus on material science, specifically the development of SMS (Spunbond Meltblown Spunbond) fabrics that offer high hydrostatic pressure resistance while remaining breathable—a critical factor for healthcare workers during extended shifts. Furthermore, the industry is seeing a move toward "smart manufacturing," where automated production lines reduce human error, ensuring that every seam of an isolation gown provides the same level of integrity.
As global health systems face increasing pressure to optimize resources, the demand for suppliers who can provide "value-added" solutions has spiked. This involves offering products that are not only affordable but also fully certified to meet international benchmarks. In this maturing market, the distinction between a generic supplier and a professional one lies in their ability to anticipate regulatory shifts and invest in the necessary quality infrastructure to meet those changes before they become mandatory.
The Significance of CE MDR Class I Approval in a Regulated Market
In the realm of international medical trade, the European Union’s Medical Device Regulation (MDR) represents the highest echelon of safety oversight. For a manufacturer like CHONGJEN, maintaining CE MDR Class I approval is a fundamental component of its global quality strategy. This regulation, which replaced the older Medical Device Directive (MDD), was introduced to enhance the clinical safety of medical devices and to provide a more transparent framework for both patients and healthcare providers.
Securing CE MDR Class I approval for isolation gowns is a rigorous process that goes far beyond simple self-declaration. It requires the manufacturer to maintain a comprehensive Quality Management System (QMS) and to produce exhaustive technical documentation. This documentation must prove the product's biocompatibility, its resistance to liquid penetration, and its structural durability under stress. The MDR also places a heavy emphasis on post-market surveillance (PMS). This means that a professional supplier must actively monitor the performance of their gowns in the field and have a systematic approach to addressing any feedback or potential risks.
For global distributors and hospital procurement officers, the CE MDR mark serves as a "safety passport." It guarantees that the isolation gowns have undergone a thorough conformity assessment and that the manufacturer adheres to the latest European safety, health, and environmental protection requirements. This level of certification is critical in mitigating legal and clinical risks for healthcare providers, ensuring that the gowns act as a reliable barrier against infection, exactly as specified in their technical data sheets. By prioritizing this approval, the company demonstrates its commitment to the highest international standards, providing peace of mind to its global clientele.
Core Strategic Advantages, Product Applications, and Client Success
Shanghai CHONGJEN Industry Co., Ltd. has built its reputation on a foundation of professional integrity and manufacturing excellence. Founded in 2014 and headquartered in the global logistics hub of Shanghai, the company leverages its strategic location to manage an efficient export network that reaches over 20 countries, including major markets in Europe, the United States, and the Middle East.
Core Advantages of the Organization:
Integrated Supply Chain: The company manages the entire process from material selection to final export. This vertical integration allows for strict control over the quality of the non-woven fabrics and ensures that the final product meets the intended protection level.
Consistency and Quality Control: Adhering to ISO 13485 standards, the manufacturing facilities utilize advanced machinery to ensure consistent output. Every batch of isolation gowns undergoes a multi-stage inspection process to verify seam strength, fluid repellency, and sizing accuracy.
Professional Expertise: With over a decade of experience in the healthcare and hygiene sector, the team provides specialized consulting to help clients choose the right material—whether it be PP, SMS, or CPE—based on the specific risk profiles of their environment.
Product Application Scenarios: CHONGJEN’s product portfolio is designed for versatility. While its CE MDR-approved isolation gowns are staples in hospital wards and surgical clinics, they are also widely used in:
Laboratory and Research: Providing a barrier against biological contaminants.
Food Service and Processing: Ensuring hygiene and preventing cross-contamination in production lines.
Industrial Cleanrooms: Protecting sensitive electronics or pharmaceuticals from human-derived particulates.
Homecare and Beauty: Offering lightweight, disposable protection for caregivers and aesthetic professionals.
Major Client Case Studies and Market Impact: The company’s primary clients include large-scale medical distribution groups and industrial supply chains that require high-volume, consistent delivery. By providing a "total solution" that includes not only gowns but also gloves, masks, and shoe covers, the company simplifies the procurement process for its partners. These long-term collaborations are built on the company's ability to maintain stable pricing and reliable lead times, even during periods of global supply chain volatility. The organization’s focus on building sustainable, long-term cooperation has made it a preferred partner for those seeking a balance of "Professional Chinese" manufacturing and "Global Standard" quality.
Conclusion
In an era where the integrity of protective equipment can directly impact public health outcomes, the role of a professional supplier is more critical than ever. Through its unwavering dedication to quality, its strategic alignment with CE MDR Class I regulations, and its deep understanding of the global PPE market, CHONGJEN continues to lead as a reliable manufacturer from China. By combining technical expertise with a customer-centric approach, the company ensures that its isolation gowns and other protective solutions meet the evolving needs of the modern world. As the industry moves toward a future defined by stricter regulations and higher performance expectations, CHONGJEN remains committed to protecting those who protect others.
For more information on product specifications and to explore the full range of healthcare solutions, please visit: https://www.chongjen.com/
Media Contact
Shanghai CHONGJEN Industry Co., Ltd. info@chongjen.com https://www.chongjen.com/