Boston, Massachusetts May 15, 2026 (Issuewire.com) - Executive Summary: Selecting the right crystal form is crucial for the performance and stability of pharmaceuticals and advanced materials. Traditional experimental screening is slow, costly, and can miss critical forms. XtalGazer™ CSP platform (website:https://www.xtalgazer.com/computational-services/#csp) addresses this by employing a first-principles quantum physics and AI-driven workflow to predict all thermodynamically stable crystal structures. This guide provides a step-by-step, hands-on implementation process—from project setup to risk assessment—enabling researchers to make faster, more confident decisions, accelerate timelines by 25%-50%, and de-risk development.
Introduction: The Critical Role of CSP in Modern solid state R&D
The solid-state form of a material—be it a specific polymorph, salt, cocrystal, or hydrate/solvate—profoundly dictates its physical and chemical properties. For pharmaceuticals, this directly impacts critical attributes like solubility, stability, dissolution rate, and ultimately, bioavailability and therapeutic efficacy. In material science, crystal structure determines mechanical strength, electrical conductivity, and optical properties. Selecting the optimal form is therefore not merely a technical step but a foundational business and development decision.
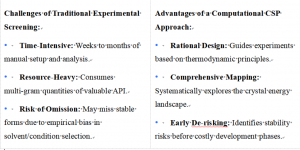
Conventional approaches rely heavily on experimental screening, such as high-throughput crystallization. While valuable, this method presents significant limitations: it is inherently time-consuming, requires substantial quantities of active pharmaceutical ingredient (API), incurs high operational costs, and critically, may fail to discover the most thermodynamically stable form. Industry data suggests that for approximately 15%-45% of marketed drugs, the most stable polymorph has not yet been identified. This gap represents a latent risk of late-stage form conversion, potentially leading to product recalls and significant financial loss.
This paradigm is shifting with the advent of advanced Crystal Structure Prediction (CSP). By moving from a "trial-and-error" to a "compute-first, validate-later" workflow, CSP enables rational, efficient, and de-risked solid-form selection. XtalPi's CSP platform stands at the forefront of this transformation, offering a robust, AI-enhanced solution to predict, analyze, and validate crystal structures with industry-leading accuracy.
Understanding Crystal Structure Prediction: Principles and Scope
Crystal Structure Prediction is a computational technique that performs a global search across defined crystallographic space to identify all energetically feasible packing arrangements for a given molecule or set of components. The core objective is to generate a ranked list of predicted structures ordered by their relative thermodynamic stability, providing a comprehensive "crystal energy landscape."
The methodology integrates two powerful approaches. It is grounded in first-principles quantum physics, primarily using Density Functional Theory (DFT), to calculate accurate lattice energies. This is combined with artificial intelligence and advanced sampling algorithms (e.g., genetic algorithms, random packing) to efficiently navigate the vast combinatorial space of possible structures. This hybrid strategy ensures both the precision of the energy ranking and the breadth of the search.
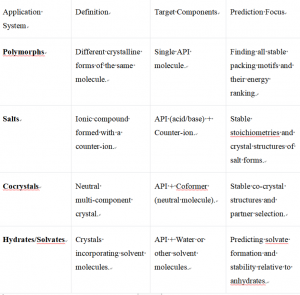
The predictive power of CSP extends across various solid-state systems critical to pharmaceutical and material development:
The value of a CSP study is twofold. It can guide the discovery of novel, previously unknown crystal forms. Equally importantly, it provides a thermodynamic context for experimentally obtained forms, cross-validating their stability order and offering confidence that the most stable form has been identified.
XtalPi's platform is distinguished by its proven performance. The underlying technology has emerged as one of the top two performers among 28 global teams in the 7th CSP Blind Test organized by the Cambridge Crystallographic Data Centre (CCDC) (link: https://www.xtalgazer.com/xtalpi-crowned-champion-at-global-crystal-structure-prediction-challenge/), and the service delivers a 98% prediction accuracy rate for covering all crystalline forms obtainable through experiments. This high success rate is powered by the synergy of proprietary AI algorithms and massive, cloud-orchestrated computational resources.
Step-by-Step Implementation Guide
This section provides a detailed, actionable four-step process for executing a CSP project with XtalPi's XtalGazer™ platform (website: https://www.xtalgazer.com/).
Step 1: Project Initialization and Input Preparation
A well-defined start is critical for project success. The primary input can be provided in one of two ways:
- A 2D molecular structure file(e.g., SMILES string, MOL file) of the API.
- API powder samples(milligram quantity), from which the structure can be determined if unknown.
The next key decision is defining the search space based on the R&D objective. This involves selecting the target system type (e.g., polymorph screen vs. salt screening with a specific counter-ion list). Collaboration with XtalPi's computational experts is recommended to set optimal parameters, including the range of space groups to explore (typically up to 230), and relevant temperature or pressure conditions for the final application.
Pre-Project Checklist:
- Confirm accuracy of the API's 2D molecular structure.
- Define the primary goal: novel form discovery, stability ranking, or risk assessment.
- Gather any existing experimental data (e.g., XRPD patterns of known forms) for future validation.
Step 2: Crystal Structure Search and Energy Calculation
Once parameters are set, the platform's automated workflow begins. Advanced global search algorithms generate a vast population of candidate crystal packings within the defined constraints. These candidate structures then undergo rigorous energy evaluation.
The most computationally intensive and critical phase is the DFT energy calculation and optimization. This refines the geometries and provides highly accurate relative lattice energies, forming the basis for the stability ranking. The entire computational process is managed via XtalPi's cloud infrastructure.
Delivery timelines are project-dependent: 2-3 weeks for routine polymorph systems, and 6-8 weeks for more complex multi-component or flexible molecule systems.
Step 3: Results Analysis and Experimental Validation
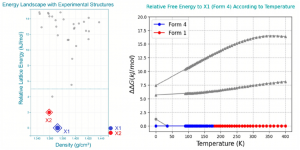
The core deliverable is the crystal energy landscape, often visualized as a plot of calculated lattice energy versus crystal density. The focus is on the low-energy region, where clusters of structures represent plausible polymorphs. The relative energy differences (often just a few kJ/mol) determine the predicted stability order.
This computational output must be integrated with experiment. Predicted X-ray powder diffraction (XRPD) patterns for low-energy structures are compared with patterns from any experimentally obtained forms. This cross-validation confirms the identity and thermodynamic ranking of known forms.
For novel predicted structures, or when traditional single-crystal X-ray diffraction (SCXRD) fails due to poor crystal quality, XtalPi's MicroED platform (website:https://www.xtalgazer.com/experimental-services/#crystal_structure_determination) provides a powerful solution. Using sub-milligram amounts of microcrystalline powder, MicroED can determine high-resolution crystal structures, with turnaround times as fast as one day. This creates a rapid validation loop, turning predictions into confirmed structural knowledge.
Step 4: Risk Assessment and Decision Making
The comprehensive energy landscape enables proactive risk management. Small energy gaps (< ~1-2 kJ/mol) between forms suggest a risk of polymorphic inter-conversion under certain processing or storage conditions, a phenomenon behind notorious cases of "disappearing polymorphs." Identifying this risk early allows for the development of mitigation strategies.
The atomic-level structural models of predicted stable forms can guide experimental chemists in selecting promising crystallization conditions (e.g., solvent classes, temperature gradients) to target specific forms, increasing the efficiency of experimental follow-up.
Finally, the structural information on novel, predicted stable forms provides a basis for strategic intellectual property (IP) planning. Early knowledge of viable new forms allows teams to evaluate patentability and design a robust IP protection strategy, securing competitive advantage.
Key Applications and Case Studies
- Energy Ranking and Cross-Validation of Experimental Forms
A primary application is determining the relative stability order of crystals already obtained in the lab. By calculating the lattice energies of all predicted forms and matching them to experimental XRPD data, CSP provides definitive thermodynamic context. This answers the critical question: "Is our chosen form the most stable one, or is there a risk of a more stable form appearing later?"
- Guided Discovery of Novel Crystal FormsCSP can directly lead to the identification and subsequent experimental preparation of new, valuable solid forms that were missed by conventional screens.
Case Study: Paxlovid (Nirmatrelvir)During the urgent development of Pfizer's COVID-19 oral antiviral PAXLOVID, XtalPi collaborated closely with Pfizer's scientists. Utilizing its CSP platform, XtalPi helped confirm the advantageous pharmaceutical crystal form of nirmatrelvir. This computational-experimental synergy contributed to significantly accelerating the development timeline, enabling faster regulatory approval and deployment of this critical therapy.
(link: https://www.xtalgazer.com/tale-of-two-polymorphs-investigating-the-structural-differences-and-dynamic-relationship-between-nirmatrelvir-solid-forms-paxlovid-2/)
- Form Conversion Risk Assessment
The complete energy landscape allows scientists to model phase diagrams and predict the stability of forms under different temperatures and relative humidities. This is vital for assessing the risk of a form converting during manufacturing (e.g., drying, milling) or long-term storage, preventing costly late-stage failures.
- Early-Stage Property Prediction
Once a crystal structure is predicted or known, computational methods can estimate key properties critical for downstream development. This includes intrinsic solubility, crystal morphology (habit) which affects filtration and flow, and mechanical properties relevant to tablet compression.
Technical Advantages of XtalGazer™ CSP
The XtalGazer™ CSP platform (website:https://www.xtalgazer.com/computational-services/#csp) delivers tangible benefits that translate directly into R&D efficiency and robustness.
- High Success Rate: The platform is designed to cover all crystalline forms that can be obtained through crystallization experiments, providing a near-complete picture of the solid-form landscape.
- Accelerated Timelines: By using AI to guide the search and optimizing computational workflows, XtalGazer™ CSP can improve project delivery speed by 25% to 50% compared to traditional or less optimized computational approaches.
- Cost and Material Efficiency: A CSP study requires no physical API samples for the prediction phase and eliminates the need for dedicated experimental screening facilities at this stage, drastically reducing material and operational costs.
- Comprehensive Scope: The service is not limited to simple polymorphs. It encompasses a wide range of systems, including salts, cocrystals, hydrates, and solvates, making it a versatile tool for comprehensive solid-state research.
Practical Tips for Success
Maximizing the value of a CSP project involves strategic planning and effective collaboration.
Project Preparation:
- Engage computational and solid-state chemistry experts early in the molecule's development timeline.
- Clearly articulate the key questions: Is the goal to find a new form, validate the stability of an existing form, or assess the risk of a known competitor's form?
- Secure and organize all relevant existing data (synthetic route impurities, preliminary XRPD, thermal data) to provide context to the computational team.
Parameter Selection & Collaboration:
- Leverage the experience of platform experts to balance search breadth (number of space groups, conformers) with computational cost. A targeted search informed by chemical insight is often more efficient than an indiscriminate brute-force approach.
- Discuss the relevance of environmental conditions (e.g., room temperature vs. body temperature for a drug) to ensure the calculated energy rankings are meaningful for the application.
Results Interpretation:
- Focus analysis on the lowest energy cluster of structures; high-energy predictions are typically not experimentally relevant.
- Understand that energy differences of less than 1 kJ/moloften lie within the computational margin of error and suggest the possibility of concomitant polymorphs or temperature-dependent stability.
- Always integrate computational predictions with experimental feasibilityand chemical intuition; the lowest-energy structure is not always the easiest or most practical to manufacture at scale.
Conclusion: Embracing the New Paradigm of Computational-Driven Discovery
Crystal Structure Prediction technology is fundamentally reshaping the approach to solid-form research in materials science and pharmaceuticals. It enables a transition from a reactive, empirical paradigm to a proactive, rational design paradigm. The "compute-first, experiment-smart" workflow powered by platforms like XtalGazer™ CSP delivers unambiguous advantages: dramatically increased efficiency, reduced consumption of precious API, and most importantly, the early identification and mitigation of stability risks that could derail a project years later.
The future of discovery lies in the seamless integration of computational power and experimental validation. By providing a complete, thermodynamically rigorous map of the crystal landscape, CSP empowers scientists to make faster, more confident decisions. It transforms uncertainty into strategy, accelerating the journey of innovative molecules and materials from concept to reality.
For more information, visit: https://www.xtalgazer.com/
Media Contact
XtalPi bd@xtalpi.com